Amanda Charlesworth, Ph.D.
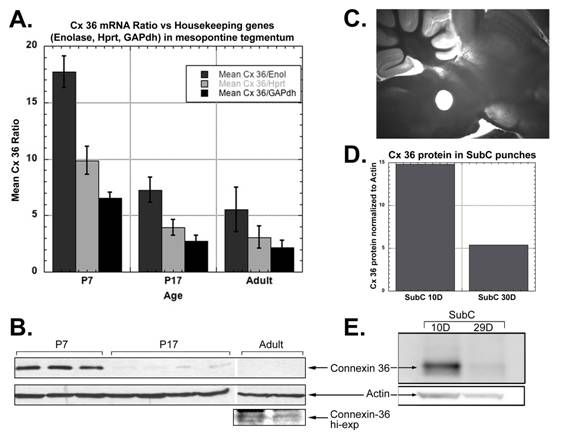
I am interested in the molecular changes that drive developmental processes. In the CTN, I am investigating the molecular mechanisms that control the developmental decrease in REM sleep. Newborn mammals spend a large proportion of time in REM sleep and by adulthood, this has substantially decreased. The reticular activating system is thought to regulate REM sleep but it is not known how. We have been investigating the hypothesis that neurons in the reticular activating system are electrically coupled via gap junctions. We have found that the neuronal gap junction protein, connexin-36, is expressed in the SubCoeruleus nucleus of the reticular activating system (see figure below). Additionally, connexin-36 levels decrease during development, a decrease that mirrors the decrease in REM sleep. The mechanisms behind the decrease in expression of connexin-36 are being investigated.
Selected Publications
Charlesworth, A., Welk, J. and MacNicol, A. M. (2000) The temporal control of Wee1 mRNA translation during Xenopus oocyte maturation is regulated by cytoplasmic polyadenylation elements within the 3′-untranslated region. Dev Biol 227, 706-719.
Charlesworth, A., Ridge, J. A., King, L. A., MacNicol, M. C. and MacNicol, A. M. (2002) A novel regulatory element determines the timing of Mos mRNA translation during Xenopus oocyte maturation. Embo J 21, 2798-2806.
Charlesworth, A., Wilczynska, A., Thampi, P., Cox, L. L. and Macnicol, A. M. (2006) Musashi regulates the temporal order of mRNA translation during Xenopus oocyte maturation. Embo J 25, 2792-2801.
Heister, D. S., Hayar, A., Charlesworth, A., Yates, C., Zhou, Y. H. and Garcia-Rill, E. (2007) Evidence for Electrical Coupling in the SubCoeruleus (SubC) Nucleus. J Neurophysiol 97, 3142-3147.
Garcia-Rill, E., Heister, D. S., Ye, M., Charlesworth, A. and Hayar, A. (2007) Electrical coupling: novel mechanism for sleep-wake control. Sleep 30, 1405-1414.
Wang, Y. Y., Charlesworth, A., Byrd, S. M., Gregerson, R., MacNicol, M. C. and MacNicol, A. M. (2008) A novel mRNA 3′ untranslated region translational control sequence regulates Xenopus Wee1 mRNA translation. Dev Biol 317, 454-466.
Yates, C., Charlesworth, A., Allen, S. R., Reese, N. B., Skinner, R. D. and Garcia-Rill, E. (2008) The onset of hyperreflexia in the rat following complete spinal cord transection. Spinal Cord
Yates, C. C., Charlesworth, A., Reese, N. B., Skinner, R. D. and Garcia-Rill, E. (2008) The effects of passive exercise therapy initiated prior to or after the development of hyperreflexia following spinal transection. Exp Neurol 213, 405-409.