Professor
Director, Graduate Program in Interdisciplinary Biomedical Sciences
Research Interest: Pathogenesis of mammalian reovirus
Ph.D.: University of Wisconsin-Madison, Madison, WI
Postdoctoral Fellowship: Vanderbilt University, Nashville, TN
Phone: 501-686-5189
Fax: 501-686-5359
Email: KWBoehme@uams.edu
Research Description

My laboratory seeks to uncover mechanisms of viral pathogenesis. We use mammalian orthoreovirus (reovirus) as an experimental system to define viral and host determinants that govern viral diseases. Reoviruses are non-enveloped viruses with genomes consisting of 10 segments of dsRNA (right). Our research is built on three focus areas. First, we are interested in determining how reovirus spreads within the host via the bloodstream. Reoviruses infect their hosts via gastrointestinal or pulmonary routes and traffic to the bloodstream which allows the virus to access every organ system in the body, including the central nervous system. We seek to understand how reoviruses access the bloodstream and move throughout the host to target organs where it causes disease. Second, we are working to define how reovirus activates and resists type-I interferon responses. Type-I interferons are a component of the innate immune system that are critical for control and clearance of all viral infections. We use reoviruses as a model to uncover how viruses activate type-I interferon responses, as well as how they overcome these responses to replicate efficiently. Finally, we have a focus on understanding how reoviruses produce their proteins in infected cells. Cell employ a variety strategies to prevent viruses from co-opting the host translational machinery to preferentially synthesize viral proteins instead of cellular proteins. Our work tries to determine how reoviruses overcome these defenses to generate the viral proteins needed for productive viral replication, which ultimately leads to viral disease.
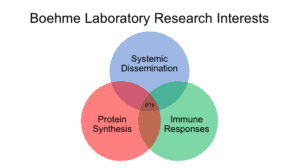
We have identified a key role for one of the reovirus nonstructural proteins, sigma 1s, in each of these three focus areas (by non-structural protein, we mean a protein that it is not part of the virus particle, not that it doesn’t have a structure). Ongoing work in the lab combines genetic, biochemical, and cell biological approaches to delineate how sigma 1s promotes viral protein synthesis and facilitates resistance to type-I interferon. We employ a small animal model that allows us to determine how our findings in cultured cells impact reovirus pathogenesis.
Current Lab Members
- Lindsey Hazeslip, MD/PhD Student
Alumni
- Michael Eledge, Ph.D. (Graduate Student) – Postdoctoral Fellow, Baylor
- Marcelle Dina Zita, Ph.D. (Graduate Student) – Postdoctoral Fellow, Johns Hopkins University
- Matthew Phillips, Ph.D. (Graduate Student) – Postdoctoral Fellow, University of Florida (Karst Lab)
- Johnasha Stuart, Ph.D. (Graduate Student) – Postdoctoral Fellow, Emory University (Grakoui Lab)
- Ryan Mann (Research Technician) – Research Technician, UAMS
- Ani Paredes (Research Technician)
- Emily Simon, M.D. (Research Technician) – Family Medicine Resident, UAMS Northeast Medical Center
- Joseph Koon (Research Technician) – UAMS College of Medicine
- Morgan Howells (Research Technician) – UAMS College of Medicine
Publications
1. Dina Zita, M., Eledge, M.R., and Boehme, K.W. Reovirus: Friend and Foe. Current Clinical Microbiology Reports. 6:132-138, 2019. doi: 10.1007/s40588-019-00121-8.
2. Phillips, M.B., Dina Zita, M., Howells, M.A., Weinkopff, T., and Boehme, K.W. Lymphatic type-1 interferon responses are critical for controls of systemic reovirus dissemination. Journal of Virology. 95: e02167-20. doi: 10.1128/JVI.02167-20, 2021. *Selected as Journal of Virology Spotlight Article.
3. Arthur, J.M.*, Forrest, J.C.*, Boehme, K.W.*, Kennedy, J.L., Owens, S.M., Herzog, C., Liu, J., and Harville, T.O.* Development of ACE2 autoantibodies after SARS-CoV-2 infection. PLoS One. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0257016, 2020*Co-corresponding authors.
4. Dina Zita, M., Phillips, M.B., Stuart, J.D., Kumarapeli, A.R., Snyder, A.J., Paredes, A., Sridharan, V., Boerma, M., Danthi, P., and Boehme, K.W. The M2 gene is a determinant of reovirus myocarditis. 96 Journal of Virology. 96: e01879-21. doi: 10.1128/JVI.01879-21, 2022.
5. Boehme, K.W., Kennedy, J.L., Snowden, J., Owens, S.M., Kouassi, M., Mann, R.L., Paredes, A., Putt, C., James, L., Jin, J., Du, R., James, L., Kirkpatrick, C., Modi, Z., Caid, K., Zohoori, N., Kothari, A., Boyanton, B. Jr.,and Forrest, J.C. Estimated pediatric SARS-CoV-2 seroprevalence in Arkansas over the first year of the COVID-19 pandemic. Journal of Pediatric Infectious Diseases. doi: 10.1093/jpids/piac010, 2022.