Assistant Professor
Research Interest: Manipulating the glycans that surround and protect bacteria
Ph.D.: The University of Iowa
Postdoctoral: University of Arkansas for Medical Sciences
Lab: Biomed I, Room 511
Phone: 501-686-7706
Fax: 501-686-5359
Email: MAJorgenson@uams.edu
Currently accepting new students
Current Lab Members
Joseph Bryant, M.S. (Research Associate III)
Emily Robbs, M.S. (Ph.D. Student)

Research Description
The Jorgenson lab is focused on investigating the bacterial surface and finding new ways to exploit its properties.
How do bacteria coat themselves? Bacteria surround themselves in sugary layers that protect against environmental hazards, antibiotics, and immune systems. Though formidable, these protective layers harbor a critical weakness: their fates are tied to a vulnerable lipid carrier known as undecaprenyl phosphate (Und-P). We are currently focused on (i) understanding how Und-P is distributed between competing pathways, (ii) defining pathways that are connected to Und-P metabolism, and (iii) exploiting Und-P production for useful purposes. By developing a deeper understanding of Und-P, the more rational and sophisticated our antibacterial therapies will become.
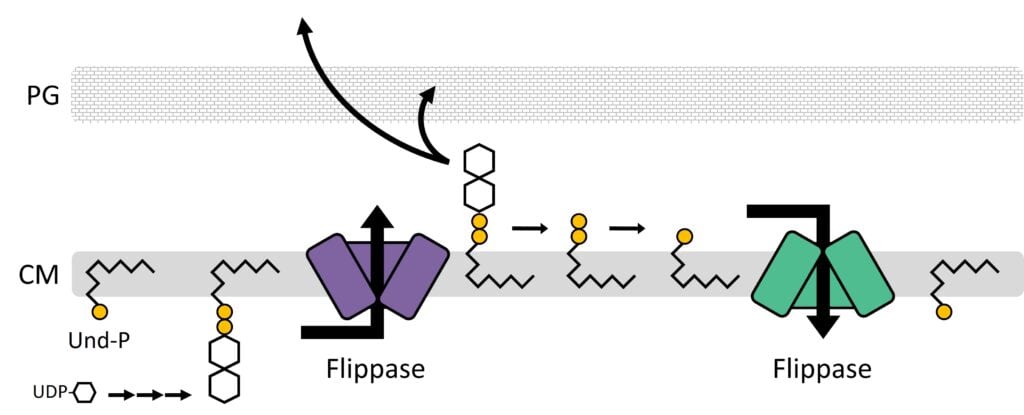
How are bacteria shaped? Bacteria are encased by a rigid superstructure known as the peptidoglycan cell wall. The cell wall confers shape and is essential to protect bacteria against osmotically induced lysis. While antibiotics target nearly every stage in cell wall assembly, the emergence of antibiotic resistance means that new targets are needed. We are currently focused on characterizing several new morphological pathways that are required to maintain cell wall integrity, as well as searching for more.
How do we address these questions? To determine how bacteria assemble and maintain their protective layers, we employ genetic, microscopic, and biochemical tools in the model organism Escherichia coli.
Publications
- Kay EJ, Dooda MK, Bryant JC, Reid AJ, Wren BW, Troutman JM and Jorgenson MA. 2024. Engineering Escherichia coli for increased Und-P availability leads to material improvements in glycan expression technology. Microbial Cell Factories 23:72.
- Jorgenson MA and Bryant JC. 2021. A genetic screen to identify factors affected by undecaprenyl phosphate recycling uncovers novel connections to morphogenesis in Escherichia coli. Molecular Microbiology 115(2):191-207. PMID: 32979869.
- Jorgenson MA, MacCain WJ, Meberg BM, Kannan S, Bryant JC, Young KD. 2019. Simultaneously inhibiting undecaprenyl phosphate production and peptidoglycan synthases promotes rapid lysis in Escherichia coli. Molecular Microbiology 112(1):233-248. PMCID: PMC6616000.
- Jorgenson MA, Young KD. 2018. YtfB, an OapA domain-containing protein, is a new cell division protein in Escherichia coli. Journal of Bacteriology 200(13). PMCID: PMC5996693.
- Jorgenson MA, Young KD. 2016. Interrupting biosynthesis of O antigen or the lipopolysaccharide core produces morphological defects in Escherichia coli by sequestering undecaprenyl phosphate. Journal of Bacteriology 198 (22):3070-3079. PMCID: PMC5075036.
- Jorgenson MA, Kannan S, Laubacher ME, Young KD. 2016. Dead-end intermediates in the enterobacterial common antigen pathway induce morphological defects in Escherichia coli by competing for undecaprenyl phosphate. Molecular Microbiology 100(1):1-14. PMCID: PMC4845916.
- Yahashiri A, Jorgenson MA, Weiss DS. 2015. Bacterial SPOR domain are recruited to septal peptidoglycan by binding to glycan strands that lack stem peptides. Proceedings of the National Academy of Sciences USA 112:11347-11352.
- Jorgenson MA, Chen Y, Yahashiri A, Popham DL, Weiss DS. 2014. The bacterial septal ring protein RlpA is a lytic transglycosylase that contributes to rod shape and daughter cell separation in Pseudomonas aeruginosa. Molecular Microbiology 93(1): 113-128. PMCID: PMC4086221.